Certainly when the almost urine got to distal tubule 85% of the water had been already reabsorbed and all that had to be done was to finish up this process. In fact only 1% was supposed to go beyond the rather uniform looking cells of the distal tubule. Gradually, however, our views began to change and through the advances in physiology, biochemistry and histology a different picture began to emerge. Nobody could have dreamed of the variety of cells in both the distal tubule and the collecting system. In a healthy person or animal, things seemed simple indeed, but let a defect or two creep in and then we begin to see the dragons. We already understand quite a bit about what can happen. If we go back one page (to page 11) we note that the very first part of the distal tubule has cells with the characteristics of those of the thick ascending loop of Henle. These cells are marked in yellow and include the macula densa. A defect in these cells leads to Bartters syndrome. Next, come the cells (in brown) that transport sodium and chloride from the lumen of the distal tubule into its lining cells by a special transporter. If the gene for the transporter is defective the end result is Gitelmans syndrome. So, what if a little salt is lost? Just a few shakes of the salt shaker should fix that but the real dragons now emerge. For reasons that we still dont understand, magnesium is poured out and calcium is retained. Our first concern was Bartters syndrome. When the first cases were described it seemed to involve little ions like sodium, potassium and chloride, substances that were regulated mainly by the renin-angiotensin-aldosterone system (RAAS). Then it was found that many Bartters patients lost magnesium and some even lost calcium as well as the expected sodium, potassium and chloride. How can we explain this? In Bartters syndrome (and in Gitelmans) the RAAS is activated but this system has nothing to do with calcium or magnesium. As to what is happening we can still only theorize. All these lost ions, Na, K, Cl, and often Mg and sometimes Ca, along with large volumes of water, travel down into the collecting system only to create chaos. A normal collecting system is now inundated with gushing fluid it cannot possibly handle. It is something like The descent into the maelstrom.
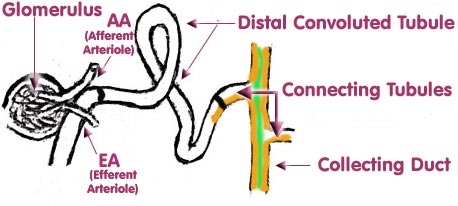
When the collecting system of the kidney is discussed, a person might say that a certain amount of double talk becomes evident. Of course, the tubular cells are absorbing glomerular filtrate from the beginning of the proximal tubule. More and more water and solutes are absorbed as the filtrate passes down the tubules. It gradually dawns that the active cells of the collecting system are those that are influenced mainly by aldosterone. This system begins in a connecting tubule (which may be of variable length) just at the end of the distal tubule. Embryologically this last part of the nephron is really not part of the nephron but is actually an outgrowth of the bladder. (Nobody is quite sure where the junction of these tissues occurs.) The diagram below, illustrating the continuity of tubular function, was taken from the diagram of the Nephron on page 3. The coloured section at the end illustrates the upper part of the collecting system.
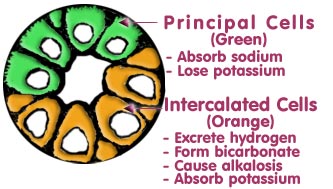
However, what really should concern us are the two, almost exclusive types of cells, lining the system. These are the INTERCALATED CELLS AND THE PRINCIPAL CELLS. They have very different functions yet these functions depend mainly on ALDOSTERONE. The intercalated cells actually begin to appear in the distal tubule (along with the cells affected in Gitelmans). These cells are marked in orange and may be seen in the connecting tubule and the collecting duct where they are much more prominent. In the collecting duct they are joined by the principal cells (marked in green) and both form the lining of the collecting duct.
Even if you are lost at this point, please read on because you will see how various drug treatments can moderate the symptoms of Bartters and Gitelmans. The intercalated cells are largely responsible for the appearance of alkalosis where the chloride is low and the bicarbonate ion is high. They accomplish this by excreting hydrogen ion. If they left a chloride behind it would be fine but unfortunately, for each hydrogen lost the blood gains a bicarbonate. So you have a lot of baking soda in your blood (except that it is ionized). High aldosterone is responsible for this reaction getting out of hand. If aldosterone is lowered the loss of hydrogen decreases and the blood becomes less alkaline. Spironolactone is generally used for this purpose but new drugs with fewer side effects are gradually coming onto the market. Please note: These cells do absorb some potassium, but are unable to cope when abnormally large amounts of potassium are presented to them in the "almost" urine from above. The principal cells also depend on aldosterone for their activity. These cells have a channel called the ENaC channel. Under the influence of aldosterone sodium passes into the cells. You will read that as the sodium goes in potassium goes out so that there is an exchange of sodium for potassium. This lowers the potassium (which is low already) even more. To keep the sodium from getting in via the ENaC channel the drug amiloride is used. Consequently this exchange is minimized. We should probably quit here and have a quaff of that kind nepenthe but here are a few more thoughts. That potassium channel in the principal cells is an ROMK channel. Remember the Rob Peter to Pay Paul channel? It is a similar channel but here there is no Paul so the potassium is lost. There are lots of things that nobody knows with regard to these channels. If you are normally healthy and are stranded on a desert island with no water, your serum sodium will go up but it will not exchange for potassium even though your aldosterone is elevated. Lyn has been informed by one who knows these things, that ROMK channels are shut most of the time. All we can hope for is more good and adequately funded research. |
||||||||
|
|